 Among patients whose tumor cells express PD-L1 greater than 1%, median DMFS was not reached with Opdivo, compared to 20.7 months with placebo (HR 0.58, 95% CI: 0.40 to 0.84). DMFS (exploratory endpoint): Across all randomized patients, median DMFS, defined as the time that patients live without cancer spreading from the primary tumor to distant organs or lymph nodes, was 47.1 months with Opdivo vs. In patients whose tumor cells express PD-L1 greater than 1%, median NUTRFS was 52.6 months with Opdivo vs. NUTRFS (secondary endpoint): In all randomized patients, those treated with Opdivo showed a median NUTRFS, defined as the time that patients lived without disease recurrence outside of the bladder, ureters or renal pelvis, of 25.9 months compared to 13.7 months for placebo (HR 0.72, 95% CI: 0.59 to 0.88). 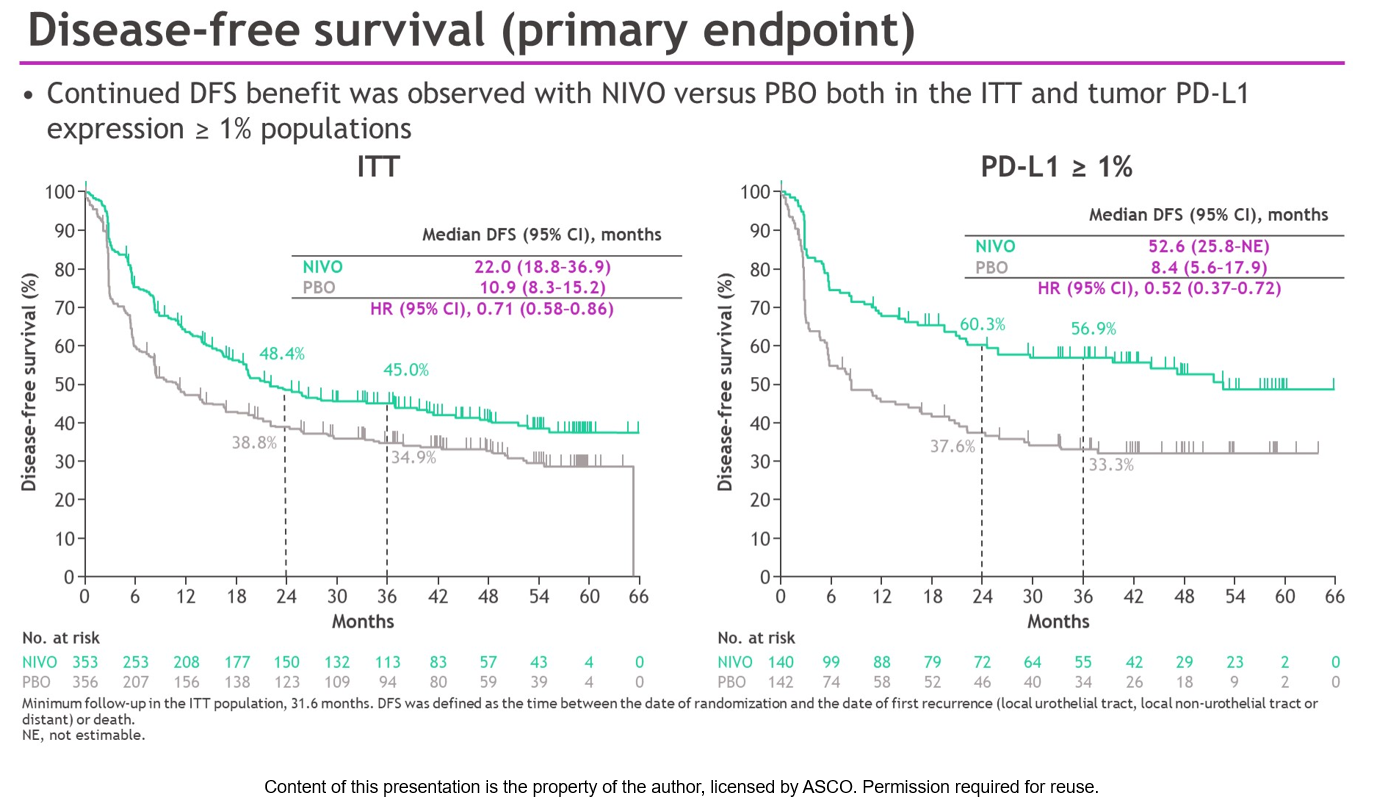
8.4 months with placebo (HR 0.52, 95% CI: 0.37 to 0.72), a 48% reduction in the risk of disease recurrence or death. In patients whose tumor cells express PD-L1 greater than 1%, Opdivo extended the average length of time patients lived without disease recurrence more than six times compared to placebo, with median DFS of 52.6 months vs. The risk reduction remained consistent with that observed at the primary analysis, with only a 1% change with an additional 25.7 months of minimum follow-up (a risk reduction of 30% was observed with 5.9 months minimum follow-up at the primary analysis HR 0.70). DFS (primary endpoint): Across all randomized patients, Opdivo more than doubled the average length of time patients lived without disease recurrence, demonstrating a median disease-free survival of 22.0 months compared to 10.9 months with placebo, a risk reduction of 29% (Hazard Ratio 0.71, 95% Confidence Interval : 0.58 to 0.86). With three years of follow-up in the CheckMate -274 trial : i. The results of this trial have changed the way that urothelial cancer is treated.” “Nivolumab remains the only immunotherapy, as well as the only medical treatment in general, to decrease the risk of urothelial cancer recurrence after radical surgery in patients who received chemotherapy prior to surgery or who are ineligible for chemotherapy. Galsky, M.D., Professor of Medicine, Director of Genitourinary Medical Oncology, Associate Director for Translational Research, and Co-Director of the Center of Excellence for Bladder Cancer at The Tisch Cancer Institute and the Icahn School of Medicine at Mount Sinai. 
The three-year results from CheckMate -274 show a stable decrease in the risk of disease with adjuvant nivolumab with longer follow-up,” said Matthew D. “Patients with muscle-invasive urothelial carcinoma face a high chance of recurrence due to micrometastatic disease, especially within the first three years after surgical removal of the bladder or kidney. These updated results will be featured in a late-breaking oral presentation at the American Society of Clinical Oncology (ASCO) 2023 Genitourinary Cancers Symposium from February 16-18, 2023. With a median follow-up of 36.1 months (31.6 months minimum), adjuvant Opdivo continued to show improved disease-free survival (DFS), non-urothelial tract recurrence-free survival (NUTRFS), distant metastasis-free survival (DMFS) and second progression-free survival (PFS2) compared to placebo across all-randomized patients and in patients whose tumor cells express PD-L1 greater than 1%. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
